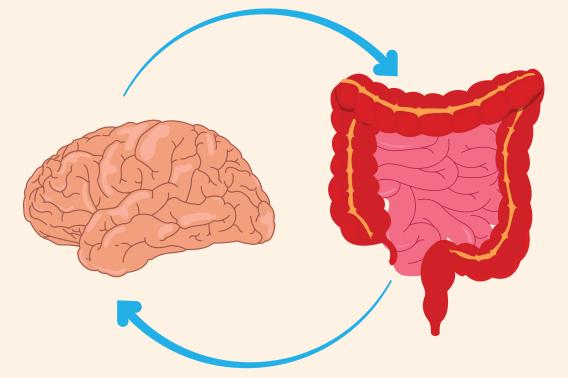
Could boosting gut–brain communication prevent memory loss?

Our memories and senses are deeply connected—like how a favorite song can recreate a whole glorious teenage summer.
It turns out this relationship might extend beyond our five external senses to include our internal senses: the signals telling us what's happening inside our bodies, sometimes beyond the veil of conscious perception.
New research by Wu Tsai Neurosciences Institute affiliate Christoph Thaiss suggests that losing these internal signals as we age — in part due to changes in our gut microbiome — could one reason why our memories decline as we get older.
Today we're talking with Thaiss—an assistant professor of pathology at Stanford Medicine and core investigator at the Arc Institute in Palo Alto—about his new study in Nature that traces a surprising path from gut microbes to memory formation in the mouse brain.
SUBSCRIBE on Apple Podcasts, Spotify, Amazon Music and more.
Learn More
- Enhancing gut-brain communication reversed cognitive decline, improved memory formation in aging mice (Stanford Medicine, 2026)
- Intestinal interoceptive dysfunction drives age-associated cognitive decline. (Nature, 2026)
- Christoph's presentation at Wu Tsai Neuro's 2025 Annual Symposium
- Neuroscientists Dive into the Gut (Wu Tsai Neuro, 2025)
- The Thaiss Lab at the Arc Institute
- Thaiss Lab publications
Episode credits
This episode was produced by Michael Osborne at 14th Street Studios, with sound design by Mark Bell . Social media strategy is by Julia Diaz, and additional editing by Nathan Collins. Our logo is by Aimee Garza. The show is hosted by Nicholas Weiler at Stanford's Wu Tsai Neurosciences Institute and supported in part by the Knight Initiative for Brain Resilience.
Share the show:
If you're enjoying our show, please take a moment to give us a review on your podcast app of choice and share this episode with your friends. That's how we grow as a show and bring the stories of the frontiers of neuroscience to a wider audience.
Get in touch:
We want to hear from YOUR neurons! Email us if you'd be willing to help out with some listener research, and we'll be in touch with some follow-up questions.
Episode Transcript
Nicholas Weiler (00:10):
This is From Our Neurons to Yours, a podcast from the Wu Tsai Neurosciences Institute at Stanford University, bringing you to the frontiers of brain science. I'm your host, Nicholas Weiler.
Our memories and our senses are tightly tied together. Like how the smell of a chocolate chip cookie might bring back memories of sitting in your grandmother's kitchen when you were seven, or how hearing the first bar of a song you used to love could recreate a whole gloriously wasted teenage summer.
But this relationship between memory and our senses might go beyond the five typical senses we usually think about; sight, smell, touch, taste, and hearing. Those are our external senses, the ways we perceive the world around us, but we also have a whole set of senses that point inward. Signals that tell us what's happening inside our own bodies.
Stop for a moment and think about how your body feels right now.
(01:08):
Where is your body in space? Are you hungry or full? Nervous, or relaxed? Is your heart beating quickly? Do you have that uneasy feeling people sometimes describe as butterflies in the stomach?
These internal sensations, sometimes called our interoceptive senses, are one of the ways the brain stays in constant communication with the body. This is something that neuroscience knows much less about than our external senses. But new research is finding these internal senses are also deeply entwined with our memories, even if those associations can be less obvious to us, or sometimes even completely unconscious.
In fact, it turns out that losing these internal signals, which happens as we get older, just like our hearing and our eyesight might start to weaken, could be related to why our memories also get cloudier as we age. And this could be a problem because while you can get reading glasses for your eyes or hearing aids for your ears, there's not much we can do, at least not yet, to repair a failing vagus nerve, for example, which is the main communication highway between the brain and the gut.
(02:16):
Today on the show, we're talking about a new study in the journal, Nature, that explores this idea in detail, tracing a path between microbes in the guts of mice all the way to the formation of memories in their brains. We'll talk about what might cause our internal senses to decline with age and how restoring them could help protect our mental abilities.
Our guest today is the study's senior author, Christoph Thaiss, an assistant professor of pathology at Stanford and a core investigator at the Arc Institute in Palo Alto. Before we got into discussing his new paper, I started by asking Christoph to tell us more about what we understand about our body's internal senses.
Christoph Thaiss (03:01):
So usually when we think about senses, we think about what we call exteroception, which is how the brain perceives the outside environment. And this largely takes place through the five canonical senses that we basically have studied for many, many decades, and we have a lot of scientific knowledge about how these five external senses work. But then in parallel, there is a separate set of senses that the brain has to figure out what's going on inside the body. Very similar to how the brain is monitoring the outside environment, it is monitoring the inside environment of the human body, but there are some very interesting distinctions between these two branches of senses. There are the five canonical senses for the outside world, we don't know if there is a finite set of senses to the inside world.
Nicholas Weiler (03:47):
You don't even know how many of these internal senses we have.
Christoph Thaiss (03:50):
That's right. We don't know how many of the variables that are inside the body in terms of the chemical milieu. We don't know how many of those are being monitored by the brain. How many of them are being monitored indirectly, and how many of them the brain ignores and are not monitored at all. But we know that there are specialized epithelial cells from the lung or in the gastrointestinal tract. They can communicate with sensory neurons, and these sensory neurons then send a signal to the brain to be understood and decoded. So there are anatomical similarities, but we don't know how many different senses of interception there are overall.
Nicholas Weiler (04:25):
Just to make sure I understand correctly. We're talking about the things that obviously come to mind are something like hunger or maybe whether I'm short of breath or I sometimes wonder whether a sudden craving for yogurt, like, I haven't had yogurt for weeks. Why do I suddenly want yogurt today? Where is that coming from? This is sort of the spectrum of different things you might imagine coming from within that where our brains are becoming aware of in some way.
Christoph Thaiss (04:51):
That's right. Everything you just mentioned are examples of consciously perceived internal sensations, but there are also many internal sensations that never gain access to conscious perception. Hunger or fullness in terms of stomach distension, these are consciously perceived internal sensations. But then there are others, if I asked you now to focus on the number of glucose molecules that are entering your gastrointestinal tract based on the last meal you just ate, even if you focused very hard, you will not be able to consciously perceive these glucose molecules. Even though we know that there are sensory neurons that innovate the gastrointestinal tract that respond to these glucose molecules. And so some of these internal sensations, they never gain access to conscious perception.
(05:36):
And then there are some interesting ones that are somewhere in between. For example, the bladder. If the bladder is full, we have to feel the bladder. There's no way not to consciously perceive your bladder if it's full. But if it's empty, we have no way of localizing our bladder. And so there are some internal sensations which seem to move from impossibility of conscious perception to mandatory conscious perception. And where this distinction is made is currently unknown, but is a very interesting aspect of interception research.
Nicholas Weiler (06:05):
So I want to talk about a specific study that you just published. Actually, it just came out today, the day that we're discussing this in March of 2026, that makes the case that something about these inner senses, or some of these inner senses, might be involved in age-related memory loss. This is a study where, as I was reading it, I kept writing, "This is wild," in the margins because there's so many interesting things here. And I'm excited first to talk about what you did and then to talk about what the heck you think it means for our understanding of memory, of aging, our internal senses, and so on. There's a lot wrapped up here.
(06:42):
So the basic discovery that we're starting with is the discovery that young mice can essentially catch memory problems from old mice. When we talk about memory, these memory losses that we see in old mice, you've got young mice and you've got old mice, you do these memory tests, and what are the differences you see in their performance?
Christoph Thaiss (07:05):
Yeah. So the memory tests, they basically, very similar to what we do in humans, they test for the ability of a mouse to remember an object that they have just seen or they remember the exit path from a maze. And so we're basically trying to test for hippocampal function because many of these learning and memory functions, both in mice and in humans are dependent on the hippocampus. And we know, of course, memory declines with age. It's extremely heterogeneous in the human population. And so actually there are some people who live to 100 of age with complete cognitive fitness and other people start becoming forgetful in their 50s and 60s. So one of the main goals in the field is to try and understand where this heterogeneity is coming from and whether there's something we can do about it.
(07:46):
So in mice, if we perform these tests, for example, the novel object recognition test is one that we use a lot in the study, the mice are exposed to different objects and then the objects are coming back or a different object is coming back and we're seeing how well the mouse remembers which object it has seen in the past.
Nicholas Weiler (08:02):
Right. And so mice tend to explore new objects. And so if you show them something, they'll explore it. If you show it to them again, if the mouse's memory is good, it won't explore it so much. If its memory is not so good, it might treat it like it was a brand new object like it's never seen it before. So this is sort of like getting at this idea of how good is the mouse's memory.
Christoph Thaiss (08:20):
Exactly. So an old mouse doesn't remember which object it has seen before, so it explores both objects, but a young mouse remembers the object very well and then spends most of the time on the new object. However, if we now co-house young and old mice in the same cage, they perform equally bad in the test. And so the really surprising discovery we made was that if we take a young and old animal and we perform memory tests on them, as we would expect, the young mouse remembers well. But then if we, instead of housing them in separate cages, we put them into the same cage, now the young animals became as forgetful as the old animals.
Nicholas Weiler (08:59):
So somehow the loss of memory with age is almost infecting the younger mice. And it sounds like from the paper, this is actually what you think is happening. And hopefully this is not something... I want to reassure listeners that they're probably not at risk from living with older relatives, because we think this is something that's happening in mice because they have this habit of eating one another's poop, their feces. There is literally something being transferred from the older mice to the younger mice. And that's sort of what set you off on this journey, right?
Christoph Thaiss (09:34):
Exactly. So when we had this observation, there were two main possibilities. One is it's a social effect that the young mice observed the old mice and then copy their behavior. Or alternatively, as you just said, it's an infectious phenomenon. So there is something about the microbes being transferred to the young mice and then the microbes are having this effect. To distinguish between these possibilities, we conducted a second experiment. This time there was no co-housing involved, but we used germ-free mice.
Nicholas Weiler (10:01):
So mice with no microbes.
Christoph Thaiss (10:03):
Exactly. They're raised without any endogenous bacteria. They're raised in sterile isolators. And so we can use these mice to ask very interesting questions about the microbiome by bringing in a microbiome of interest. And so in this study, we took microbiome samples from young and old donors, we transplanted them into the germ-free recipients and then performed the exact same memory tests on those recipients. And in these experiments, we saw that all young mice, no co-housing involved, but the recipients of an aged microbiome or a microbiome from an aged donor, they started showing signs of cognitive aging. And so we can basically, with these experiments, we can disentangle the age of the microbiome from the age of the host, and we can see that cognitive aging tracks with microbiome age rather than host age.
Nicholas Weiler (10:49):
Okay. So we observe that as is typical, older mice have worse memory. They don't recognize objects. We also did experiments with mazes where older animals had trouble getting out of the maze in a timely fashion, and that there's something in their microbiome, it seems, that is conferring this effect. Because if you get rid of the microbiome, the older animal's memories get better. And if you give the old microbiome to a young mouse, it makes the young mouse's memory worse. This raises the question, what is in the microbiome that is conferring this effect on memory? What was your next step here? Where did you take this from there?
Christoph Thaiss (11:27):
So whenever we have a question like this in the microbiome field where we have basically a microbiome of interest associated with a phenotype, in order to identify candidate species, we can use next generation sequencing. So we can identify the taxonomic members of the microbiome that would most closely associate with the phenotype. And then we can nominate candidate species by looking at, first of all, which species are different between young and old, which of them evolve over the lifespan, and which of them successfully engraft into germ-free mice or transfer in this co-housing assay. And once we have this triangulation of possibilities, then we can nominate candidate species. And in our case, the top hit was a bacterium called parabacteroides goldsteinii which is known member of the mouse and human microbiome. And we actually had some indications from the literature that other members of the parabacteroides genus might be involved in the regulation of cognitive processes. So then we got very interested in exploring these bacteria.
Nicholas Weiler (12:24):
Okay. So you identified this particular bacterium as something that is there in the mouse and the human, it increases with age in the mouse, and it's the only one that you saw was able to transfer to the young mice and that it had this effect on aging.
Christoph Thaiss (12:40):
Yeah. So I should point out it's certainly not the only one that increases with age, and it's also not the only one that is successfully transferred to young mice, but it was the only one which also had a detrimental effect on memory.
Nicholas Weiler (13:04):
Okay. So now we have a potential culprit. Something about the microbiome can get transferred from old mice to young mice, and you've identified this particular bacterium, parabacteroides, goldsteinii, that has all the fingerprints of the culprit in this situation. Which then takes you on this big journey to try to figure out, well, how do you connect a bacterium in the gut of mice to memory problems that these mice are experiencing? I wonder if we could leave the bacterium for a moment and switch over to what we think is going on in the brain, because we're going to have to make that connection at some point. I think it might be a little easier to start with the brain and work our way down.
(13:45):
So this brings us to some experiments that one of your students was doing a few years ago looking at, well, with aging, what exactly is going on in the brain? Why are these animals performing less well on these memory tasks? Can you set the scene for us here? We talked about the memory experiments. What was your student seeing in the brain that seemed to be related to the older mice's poor performance?
Christoph Thaiss (14:07):
So learning and memory, both in mice and in humans is very strongly linked to the activity of the hippocampus. And actually we know memory formation is very strongly linked to the formation of what we call memory engrams. So these are basically ensembles of neurons that get activated in response to a stimulation. For example, if I'm making a sound with my hands, the neurons in our brains are activated in response to the sound, and the ones that get co-activated will encode different aspects of this experience. And then the engram consolidates remains over time, but then if the stimulation comes back, for example, if I'm making the same sound again with my hands, now the same neurons get reactivated and produce the experience of the memory. And that's why we, for example, if we enter a room and we see a picture on the wall and we see the same picture on the wall in a different context later on, basically the remaining neurons in the engram will fill in the picture of what we have seen this picture. Basically will explain to us that we have seen this picture in the room before.
Nicholas Weiler (15:08):
Because the engram is encoding not just the thing you're seeing, but also where you were and what was going on. And so you see that picture and it suddenly brings back, "Oh yeah, I was walking with my mother in San Francisco and we had just had lunch and we walked into this museum and I saw that piece on the..." It completes this memory and that's sort of what we think of as a memory actually.
Christoph Thaiss (15:28):
Exactly. So the current understanding of these engrams is that there are different features of the experience are encoded by different neurons, but their co-activation is what produces the phenomenon of a memory. And so the reason why I bring this up is because engram formation declines with age. That's exactly what we saw in the study when we compare just young and old mice. We know that in young mice, the engrams form very strongly in response, for example, to an object stimulation. In an old mouse, it's much less the case.
(15:56):
Now, the interesting observation that it was that if we do our microbiome manipulation, now we can suppress engram formation because we can suppress the level of neuronal activation in the hippocampus. And so this suggested to us that what's going on is that the microbiome somehow is being sensed by what's going on in the hippocampus, and this has then these dramatic effects on memory encoding in the brain.
Nicholas Weiler (16:19):
And it wasn't only in the hippocampus where you saw differences in activity in the brain, right? There were also these sort of, at least to me, not being in this field, sort of surprising changes in sensory systems.
Christoph Thaiss (16:32):
Yes. We tried to see how generalizable this phenomenon is, how specific it is to the hippocampus or how many other areas of the brain are affected. And so we looked and to see whether these microbiome changes that usually occur with age are affecting the neuronal activity outside of the hippocampus. And then we very quickly saw that among the regions that are most affected, seem to be the one that are integrating sensory information. For example, the brainstem or some other sensory cortex or other areas of the brain that are well known for their role in sensory integration.
Nicholas Weiler (17:02):
So maybe there's somehow less sensory signaling from the body that might be related to the memory problems?
Christoph Thaiss (17:10):
Exactly. This is what led us to hypothesize that what is happening in aging and in the context of gastrointestinal aging specifically, is reduced sensory perception. And so as I mentioned at the beginning, we know very little about how this interoceptive system ages, but this suggested that there is a signal coming from the gastrointestinal tract, which is declining over the lifespan. And then we hypothesize that this decline in sensory perception from the gastrointestinal tract might be causally involved in brain aging and memory decline.
Nicholas Weiler (17:40):
So you didn't leave it there. You kept exploring what exactly the mechanism might be. We've got this microbe that's in the gut that we identified as potentially a culprit in memory decline, and we're seeing changes in the hippocampus in memory formation and in these sensory regions where we're seeing less sensory input from the body. So help us draw the connection there. How do we connect a microbe in the gut and changes in memory formation in the brain?
Christoph Thaiss (18:12):
So when we talk about interception, we basically talk about multiple parallel sets of neuronal cables that go from the brainstem to the different tissues of the body. So almost any tissue in the body is innovated by these neurons, and they largely fall into two categories. Either it's neurons that run along the spinal cord or it's neurons of the vagus nerve. And so we had to distinguish between these two possibilities because both spinal neurons, as well as the vagus nerve, are innovating the gastrointestinal tract.
(18:40):
So we used a few genetic tricks that we can do in mice to distinguish between these two possibilities, and all of them pointed to the vagus nerve as being the most important player. And so we hypothesized that the vagal connection between the GI tract and the brain is what leads to input into the hippocampus, but also leads to these declining signals as we get older.
Nicholas Weiler (19:00):
And this is another place where I wrote, "That's wild," in the margins. Part of those experiments where you could silence the vagus nerve and that recreates some of these memory problems in young mice. So if you block the vagus nerve, you see similar memory issues in young mice suggesting we created the same memory problem that we're seeing with aging by silencing the vagus nerve, and you were able to stimulate the vagus nerve and you share that that rescues memory in old mice. So there's something about activity of the vagus nerve that is deeply involved in this issue where we're seeing loss of memory in older mice. And you also use GLP-1, right? To stimulate the vagus nerve, is that right?
Christoph Thaiss (19:39):
Yes. Other stimulations that we can use to activate sensory neurons, such as GLP-1, but also other gut hormones like CCK, they have the same effect. So whichever means we use to stimulate sensory input into the brain, counteracted memory decline. And so it seems like this is a pretty broad effect of sensory neurons. We actually don't know exactly which subset of sensory neurons is exactly required for this phenomenon, but we know that the non-specific reactivation of these sensory neurons is able to restore hippocampal function and memory formation.
Nicholas Weiler (20:11):
Okay. So actually, I want to pause here for a moment. We've made a lot of connections and there's a few connections to make to understand exactly what's going on here. But what we have so far, there's a loss of sensory activity in the vagus nerve in older mice, and we'll get back to what is causing that. And that in some way is reducing the older mice's ability to form new memories. Why do you think that is? What is it about lower activity in this vagus nerve that would affect memories? Because I mean, we mentioned this a couple of times. We think about our external memories, our sights, our smells, our sounds, but I don't really think about my memories as being particularly internal. What is the vagus nerve doing in my memories or in a mouse's memories?
Christoph Thaiss (20:56):
Yeah, that's a really good question. I wish I had an answer to this question. It's basically asking why does the pathway even exist? What's the evolutionary origin and how does the vagal input into the brain modulate memory? So we have a few speculations, a few possible answers. One of them is that the vagus nerve serves as a contextualizing signal. So basically during human evolutionary history, it was probably very beneficial to contextualize memories through an internal signal. For example, to remember things much better if you were extremely hungry or to remember the location where you found food, if you also ate the food and it was rich in nutrients. And so the vagus nerve would relay this contextualizing signal, which can then make memories stronger or weaker depending on the contextualizing signal.
Nicholas Weiler (21:46):
It's interesting. As you were describing this idea about internal signals providing the context for memory, I don't think I have really strong memories of, for example, the feeling of being hungry in a particular situation, but I could certainly imagine that I would be much more likely to remember, for example, a meal when I was very hungry or remember being incredibly thirsty and finding water somewhere. In fact, I do have a memory of that, which is when I was young, I was in the hospital and wasn't able to drink for a long time. I still very clearly remember the first drink of cranberry juice that a kind nurse gave me after some amount of time of not being able to drink.
(22:29):
So I think that I'm getting an idea of how these internal senses, these internal states, might be influencing how well memory works. But I think it's still so fascinating that you're saying, if you don't have those internal signals, it looks like it is much harder to form these memories at all.
Christoph Thaiss (22:50):
It's actually an open question, whether we also have the ability to form memories about internal states. There is some suggestion that, for example, signals from the immune cells of the body form an engram-like state in the brain and can be reactivated by reactivating this engram. And so it's possible that there are internal sensations that give rise to memories just like external sensations would, but it's equally likely that they form this contextualizing signal that I was mentioning in order to strengthen or weaken the memories that are generated in response to outside stimuli.
Nicholas Weiler (23:22):
Okay. So we've left parabacteroides goldsteinii sort of sitting on the table waiting for us to come back to it. We are able to see that something about this memory loss can get transferred from old mice to young mice, and that you could replicate that just by transferring their microbiome, the bacteria in their guts, and that you could protect the old mice from getting this memory loss basically through raising them without any microbes. And you did a big search and figured out that the one microbe that looks like the biggest likely culprit is this microbe parabacteroides goldsteinii. So now we've got parabacteroides goldsteinii and we've got reduced activity in the vagus nerve, which is impacting memory. Help us draw the connection there. How is this microbe changing the activity in the nerve that is supposed to provide this contextual information from our guts?
Christoph Thaiss (24:16):
So to answer this question, we leveraged the observation we had that parabacteroides goldsteinii was able to have this cognitive effect, but other members of the microbiome that had similar properties in terms of changing with age and being transmissible to germ-free mice or co-house partners did not have a cognitive effect. One of these controlled bacteria was alistipes shahii, for example, which goes up with age is transmissible to the co-housing partners, but in isolation does not have a cognitive effect.
(24:43):
So now we had two sets of species, one that has a cognitive effect, the other one that does not have a cognitive effect, and we wanted to compare these two to identify where the difference is coming from. And so instead of transferring these bacteria themselves to the recipient mice, we just transferred their supernatants, basically their metabolic products.
Nicholas Weiler (25:02):
So basically they're sitting in a little broth of their own production and then you just put the broth in the animals, so not the bacteria themselves, but whatever the bacteria are producing.
Christoph Thaiss (25:10):
That's right. They sit in cultures, they produce things into the supernatant, and we transferred the supernatant and just the supernatant from parabacteroides alone was sufficient to induce cognitive decline, while the supernatant from alistipes would not have a cognitive effect. Which suggested that we actually don't need live bacteria to recapitulate this effect, but it's sufficient to look at the metabolic products. And so then we used metabolomics to just unbiasedly explore what's in these supernatants, and we found an enrichment of medium chain fatty acids.
Nicholas Weiler (25:41):
What are those?
Christoph Thaiss (25:42):
These are molecules that are either produced by bacteria. They're also very frequently found in dietary components, like in the food, but they can also be produced by cells of the host, by the human cells or by the mouse cells. And so they are very pervasive molecules and they have many, many different functions in biology, but the function that we focused on is their ability to be perceived by immune cells and then trigger inflammatory signaling. And so there is a receptor for these medium chain fatty acids, it's called GPR84, and it's known to be expressed primarily by myeloid cells, which are cells of the immune system. And the consequence of these myeloid cells sensing the medium chain fatty acids is the production of pro-inflammatory cytokines, basically mediators of inflammation.
(26:28):
So what this meant is that this change in the gastrointestinal environment leads to an accumulation of medium-chain fatty acids. And these medium-chain fatty acids are actually perceived by the immune system. So the immune system monitors these molecular changes, and in response to the molecular changes starts producing inflammatory cytokines. Now, the reason why this was interesting is because we know for a long time that there is a phenomenon called inflammaging, which is basically describing that the inflammatory tone in different tissues increases as we get older.
Nicholas Weiler (26:57):
Yeah, we had a great conversation about that with Katia Andreas and a few months back.
Christoph Thaiss (27:02):
Yes, yes. Katia is actually one of the leaders in the field of trying to identify where these inflammatory signals come from. And so our study would suggest that there is a microbial contribution to this inflammaging effect and that these medium chain fatty acids and their signaling through GPR84 might be responsible for some of the inflammaging effect. And the reason why this is interesting is because inflammaging is a major risk factor for many, many of the known age-associated diseases, metabolic diseases, neurological diseases, and many others as well.
(27:32):
So that's definitely something that we try to avoid. And we actually, we have a lot of anecdotal evidence that the lifestyle interventions that are helpful in terms of preventing age-associated diseases like frequent exercise and healthy diets, caloric restriction, intermittent fasting, all of these seem to act lowering peripheral inflammation. That's why it was an interesting observation that these microbial changes that accumulate over the lifespan seem to contribute to inflammaging in the periphery.
Nicholas Weiler (28:00):
Right. And you saw specifically that you could get rid of these inflammatory immune cells, you could get rid of the molecules, the cytokines that they're producing, and that would, again, rescue this aging thing, right? If you got rid of those things, then the aged mice had better memory and that this inflammation is specifically acting on the vagus nerve, which we've talked about. You can show that there's a specific effect of these inflammatory cytokines on the vagus nerve that's leading to this whole effect.
(28:31):
So now we have sort of the whole chain. With age, the microbiome changes, one microbe that changes is parabacteroides goldsteinii. That microbe is producing medium-chain fatty acids. There's, I think one in particular you highlighted that might be a problem. Those are triggering myeloid cells to produce inflammatory cytokines. Those are reducing the activity in the vagus nerve. That is reducing input to the sensory systems of the brain, and that is leading to an inability of the brain to form memories.
(29:05):
And that's an incredible chain from putting old and young mice together in cages to understanding why the old mice are having trouble with their memory. And one of the things I thought was so clever about this study was basically at every stage of this chain, you are identifying, well, we can turn this up and down, right? We can have this intervention and it makes memory worse and we can have this intervention and it makes memory better. And so now you have sort of a menu of options if you wanted to think about how could we translate this to finding ways to improve memory in humans. Before we get to that, I mean, I guess the question that has been on my mind is how many of these components are present in humans? Is there good reason to think that a similar pathway is present in ourselves?
Christoph Thaiss (29:54):
So strictly anatomically speaking, all the components of the pathway are certainly there. Parabacteroides, as I mentioned, is a member of the human microbiome as well. These myeloid cells in humans can certainly respond to medium-chain fatty acids. Vagal anatomy is quite well-preserved between mice and humans and obviously the involvement of the hippocampus in human memory and learning and cognitive decline has been studied for decades. So strictly from an anatomical perspective, all the components are there.
(30:23):
Whether the same pathway is involved in mediating cognitive decline in humans is something we don't know, but we're gearing up to conducting human studies that would actually allow us to tease apart some of these components in humans as well. One thing that's nice at Stanford Medicine is that we have access to many cohorts that are looking at this exact question, following individuals over the lifespan and looking at their cognitive development over time and how different environmental factors would actually modify cognitive development over time.
(30:55):
There is anecdotal evidence in the literature because vagus nerve stimulation is an FDA approved procedure, for example, in treatment refractory depression, epilepsy, and as well as motor recovery after stroke. And so there are individuals who stimulate their vagus nerve for therapeutic purposes. And there is anecdotal evidence in the literature that it actually leads to an improved memory function in these individuals, but that's something that we now need to explore in thoroughly designed trials.
Nicholas Weiler (31:23):
How would you go about doing this in humans?
Christoph Thaiss (31:25):
So what's next is that we can link changes in the GI environment with cognitive decline over the lifespan. We can leverage some of these large ongoing cohort studies and we can explore whether there are individual components of the gastrointestinal tract that would associate with the cognitive performance or memory performance of these individuals over time. But there are also interventional studies that we can think about. For example, vagus nerve stimulation, as I mentioned, is currently achieved basically using a electrical stimulation of the vagus nerve. And there's even a more science fiction component to this where we're trying to develop devices that would allow us to record the activity of the vagus nerve and would potentially tell us by proxy what's going on inside the peripheral and central nervous system of these individuals.
(32:11):
The reason why this is important, I think, is because we know a lot about what's going on in the circulating system of people. And of course, there are many studies that have linked changes in the peripheral blood to cognitive changes in older individuals. Every time we go to a physician, we have a blood draw because the blood is so informative about the activity of different peripheral organs and allow us to make diagnosis about specific diseases, for example. But we don't have the equivalent for the peripheral nervous system yet, because there are no wearable devices that would allow us to study the activity, the electrical activity of the peripheral neurons, and it's much harder to assay them. If we go to a physician's office, they would usually not stick an electrode into-
Nicholas Weiler (32:51):
Right. They can't check your vagus nerve for you at the moment.
Christoph Thaiss (32:54):
But that would be ideal to have.
Nicholas Weiler (32:55):
Right.
Christoph Thaiss (32:56):
That would be a tool that would allow us to ideally non-invasively monitor the activity of peripheral neurons and would allow us to make associations between changes in these activities and disease states. So that's maybe a little bit further into the future, but that's a future that would be very exciting.
Nicholas Weiler (33:13):
So that what happens in vagus doesn't stay in vagus. I'm sorry, I had to say it.
Christoph Thaiss (33:16):
Exactly.
Nicholas Weiler (33:35):
I mean, it does seem like one of the big takeaways here is that we need to be thinking a lot more about what the vagus nerve is doing, the communication between our bodies and our brains, and how critical it is, or appears to be, for our health. It certainly seems critical for memory in mice. I guess I've got two translational questions, which is what are things that we know activate the vagus nerve and are there any downsides to stimulating the vagus nerve, particularly if it's in a fairly mild way?
Christoph Thaiss (34:03):
So to the former question, we know that many of the sort of healthy lifestyle interventions that we know counteract age-associated disease, they actually activate the vagus nerve very strongly. So my group is very interested in understanding how exercise impacts sensory neuron input into the brain, but there's a lot of evidence that other things, including diet, strongly modulate the activity of the vagus nerve. So it's actually possible that at least some of these beneficial effects of these lifestyle interventions on cognitive health are mediated through the modulation of vagal activity, and that's something that we're actively exploring.
(34:36):
There's also, again, going a little bit further into the future, there is a possibility that we can pharmacologically remote control what's going on inside the brain by modulating the activity, not of the brain directly, but by modulating the activity of these sensory neurons. In my group, it's an approach that we call interocepto-mimetics because it's allowing us to mimic the activity of what's normally occurring in the interoceptive system, but we're trying to leverage this for therapeutic purposes. And possibly the most famous and first very impressive example of interocepto-mimetics is actually the GLP-1 receptor agonist drugs, because GLP-1, as we talked about earlier, is an endogenous interoceptive signal. It's produced by the gastrointestinal tract, but then its pharmacological overactivation is leading to all of these impressive weight loss effects and many other beneficial effects that these drugs seem to have.
(35:29):
So this is an example where we have an endogenous signal, and if we basically take the endogenous signal and we turn it into a therapeutic approach that dramatically overactivates the activity of its receptors, in sensory neurons but also in the brainstem, then this leads to all these beneficial outcomes in terms of brain health, but the health of many other tissues as well. And so the question we have is whether this is actually a generalizable principle of these efforts to activate sensory neurons that may have so many beneficial effects down the road.
(35:59):
The other thing you asked is whether there is a downside to it, and we have to be very careful because we know, as I alluded to just a minute ago, that the overactivation of some of these sensory systems may actually lead to the opposite outcome, that there is overactivation induced hyposensitivity of these neurons. And so that's certainly something we would want to avoid. But based on the experience we have with the GLP-1 receptor agonist drugs, it appears that chronic stimulation of these gut-brain pathways is certainly possible and can certainly have a beneficial outcome. What I should say though is that the GLP-1 receptor agonist drugs, they don't exclusively act on the vagus nerve because the GLP-1 receptor is expressed in other areas of the body, including in the brainstem, and it's actually believed that the brainstem is the main site of the therapeutic efficiency of these drugs. So that would suggest that sensory reactivation doesn't only mean reactivation of the vagus nerve, but it may involve other areas of the nervous system as well.
Nicholas Weiler (36:51):
But on the other hand, I mean, the main therapeutic effect of the GLP-1s, they were developed for diabetes and now they're used extensively for weight loss, but it's been documented that they have many other effects. I mean, they have effects on addiction people report or on mood and so forth. So as you say, those receptors are all over. Maybe some of those effects are coming from the vagus, maybe some of them are coming from the brainstem.
Christoph Thaiss (37:15):
Yes. That's actually a very interesting aspect of these drugs is that they certainly have many unwanted but biologically logical side effects, including nausea and vomiting, because endogenously GLP-1 is signaling too much intake into the gastrointestinal tract. If there is a lot of undigested material that arrives in the distal bowel, it will trigger GLP-1 release from enteroendocrine cells. And so the body is basically signaling to the brain that it's time to stop introducing material into the gastrointestinal tract because the gastrointestinal tract doesn't manage to keep up with digestion, or because there is undigestible material.
(37:50):
And so nausea and vomiting is actually a very logical reaction to this. And so the fact that this is a very frequently observed side effect of these drugs makes a lot of sense in terms of the biology of these drugs. Some of the other nicer side effects, including the ones you just mentioned on addiction, I think are a little bit harder to explain based on a purely biological function of GLP-1, but may actually be a result of the therapeutic overactivation that is rarely observed under physiological conditions.
Nicholas Weiler (38:17):
So just to tie a bow on all of this, we've learned so much about the importance of this gut-brain axis, the way that our guts and our brains are communicating with one another. And you've done a lot of work on this, that I'll link to in the show notes, that are helping us understand different aspects of communication, which goes both ways. I mean, the gut certainly can influence the brain, the brain can influence the gut. And just to sort of leave the audience with a takeaway, much of this is, this is fundamental biological research. We're understanding these systems and we hope to translate it to humans in the future. Does any of this change just as a human being, as well as a scientist, how you think about taking care of your own body, your microbiome, your vagus nerve, things like that? Does it alter how you move through the world or not so much yet?
Christoph Thaiss (39:05):
I have to admit that I'm not following all the latest trends and suggestions about how we can improve the health of our gastrointestinal tract, but it has sensitized my understanding of what we usually call the embodiment of intelligence, which means that the brain is certainly not this excluded organ that sits on top of the body and has nothing to do with the rest of what's going on inside the body. But there is cognitive processes like learning and memory, which are extremely fundamental and are so necessary for the human experience, they are not solely brain intrinsic, but they rely on input from the gastrointestinal tract in our study and probably many other parts of the human body as well.
(39:47):
So everything that defines me as a person, and the reason why I remember who I am and the reason why I can relate to everything that we're talking about today, is basically because I can form these memories. And I can remember my childhood, I can remember what happened yesterday. And it seems like much of this, I wouldn't say all of this, but much of this depends on me having a body, not just my brain being hooked to some computer somewhere. So that is a very important implication of these basic findings that we had.
(40:17):
If we think about artificial intelligence systems, if we think about the brain simulation efforts that are going on all over the world. And so for me as a person and as the father of young kids, when they are learning, this is certainly the case that the teachers always tell us that kids learn much better if they don't just see an object or a specific material, but if they interact with it, if they activate other sensory stimulations in response to the learning activity. And so I think this goes back to what we talked about earlier in terms of the contextualization of memory formation through sensory experiences. And I think that's something that even in modern human learning is extremely relevant.
Nicholas Weiler (40:54):
That's a beautiful place to end. Thank you so much, Christoph. It's been a pleasure talking about this research with you. I look forward to hearing more about how it goes in the future.
Christoph Thaiss (41:02):
Thanks very much for having me.
Nicholas Weiler (41:04):
Thanks again so much to our guest, Christoph Thaiss. He's an assistant professor of pathology and a core investigator at the Arc Institute in Palo Alto. To read more about his work, check out the links in the show notes.
(41:17):
If you enjoyed this episode, please subscribe to the show for more conversations from the frontiers of brain science. We also love hearing from listeners. If you have thoughts about the show or questions about the brain you'd like to hear us discuss in a future episode, send us an email. We're at neuronspodcast@stanford.edu, or you can leave us a comment on your favorite podcast platform. And while you're at it, please give us a rating and share the show with your friends. It may seem like a small thing, but it is tremendously valuable for us to allow us to bring more listeners to the frontiers of neuroscience. Coming up on From Our Neurons to Yours.
Corey Shain (41:56):
There's a kind of hidden structure behind most of the sentences that we hear and produce that we're probably not aware of. But for example, if I say Corey talked to Nick this morning, I can form a question whose answer is Nick from that statement. So who did Corey talk to this morning? But I can't form a question whose answer is morning, as what did Corey talk to Nick?
Nicholas Weiler (42:19):
From Our Neurons to Yours is produced by Michael Osborne at 14th Street Studios, with sound design by Mark Bell. Our social media strategy is by Julia Diaz, additional editing by Nathan Collins. Our logo was designed by Amy Garza. I'm your host, Nicholas Weiler. Until next time.