Newly identified chronic pain circuit offers pathways to new treatments
A new map of a brain circuit specific to chronic pain suggests a promising route to treatment for the roughly 60 million Americans living with persistent pain, according to a study published in Nature.
The study showed that silencing the specific cells that drive this circuit eased chronic pain while preserving acute pain responses—in other words, the body’s ability to signal danger.
“A surprise to us was that acute pain and chronic pain can be completely separate,” said senior author Xiaoke Chen, a Wu Tsai Neurosciences Institute affiliate and associate professor of biology at Stanford Humanities and Sciences. “There is a dedicated circuit that only activates after injury, which gives us the opportunity to target the chronic pain component but leave protective acute pain intact.”
The research was supported in part by the NeuroChoice Initiative, a Wu Tsai Neuro Big Ideas in Neuroscience project focused on understanding biological mechanisms of addiction, including the risk for people who take prescription opioids for chronic pain.
A misinterpretation in the brain
Pain is a critical response all animals have evolved to warn us of danger and motivate us to seek healing. The defining feature of chronic pain is that it persists long after the immediate threat has dissipated. It can follow an injury, inflammation or other condition, and people with chronic pain are at greater risk of mental health problems and opioid misuse.
A hallmark of chronic pain is sensitization, where patients become hypersensitive to touch and other normally benign sensations. “In chronic pain, the brain misinterprets touch to be a painful stimulus,” said Chen.
Identifying the specific neurological circuit loop responsible for this misinterpretation could help correct it. Previous research has shown it’s possible to dampen pain by electrically stimulating the periaqueductal gray (PAG) and its downstream region called the rostral ventromedial medulla (RVM). Researchers have hypothesized that the PAG-RVM may form a system that’s important for chronic pain as well, but researchers had yet to identify a full circuit for chronic pain.
Mapping a new pain pathway
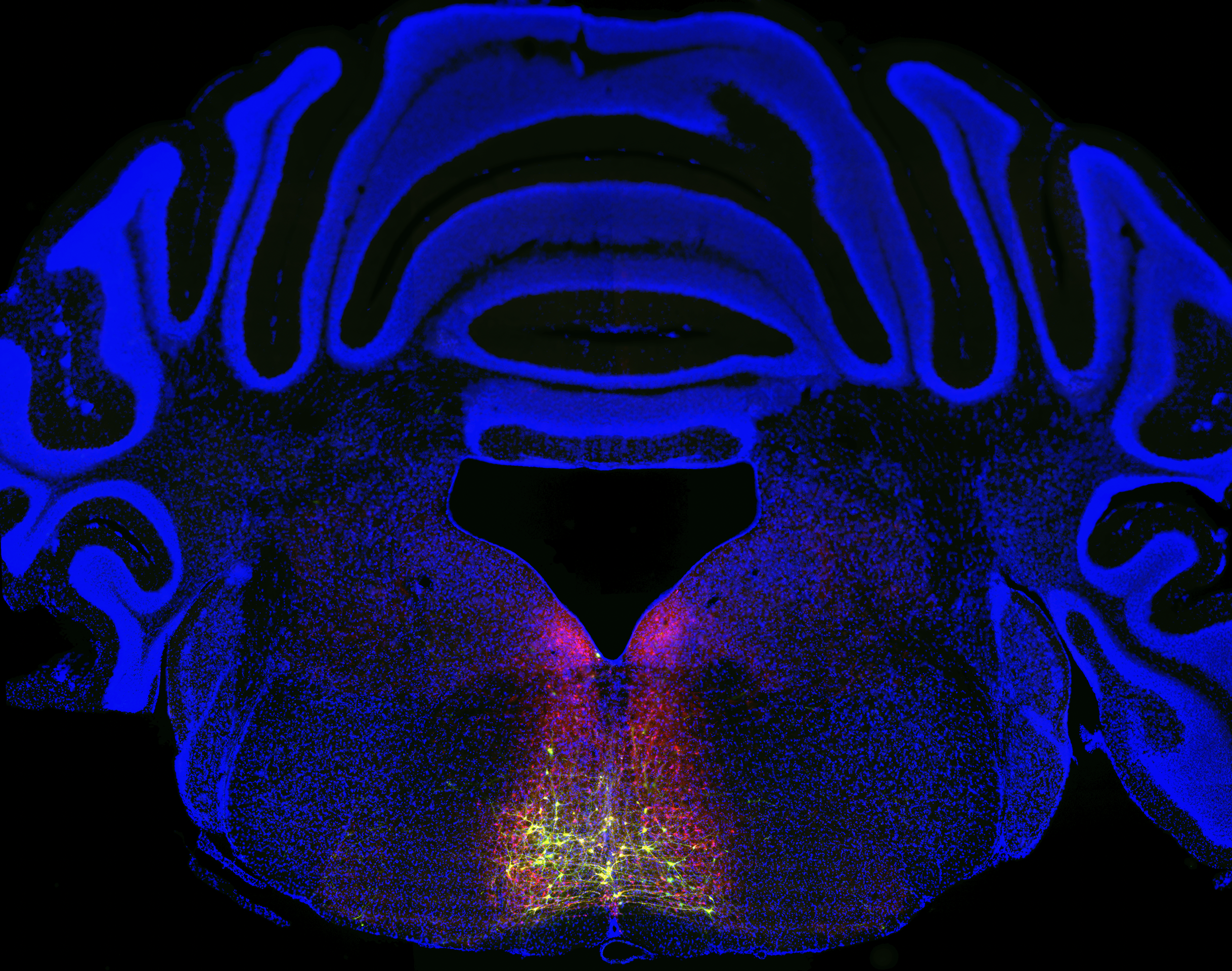
To find that full circuit, Chen’s team worked backwards from a group of neurons found in the RVM known to be involved in pain sensitization. Building on previous genetic methods, they tagged neurons along the chronic pain circuit with a fluorescent protein.
The glowing trail that resulted revealed a previously unknown circuit loop originating in the spinal cord, then linking to the thalamus, the cortex, the brainstem (where the RVM is located), and back to the spinal cord.
By chemically silencing that circuit, the team showed it was specific to chronic pain. Before silencing, mice shied away even from relatively gentle touches—a key sign they were experiencing chronic pain. Afterward, the mice responded appropriately to gentle and rougher stimuli. In other words, their chronic pain subsided, leaving their protective acute pain response intact.
“When silencing this group of cells, the sensitized pain goes away,” said Chen. “Therefore, the activity of these cells is necessary for injury or inflammation-induced pain sensitization.”
Activating the same circuit in healthy mice actually induced chronic pain. By chemically stimulating neurons in the circuit multiple times, the team found they could increase pain sensitivity in the mice and that this sensitivity lasted for weeks afterward. “Just activating these neurons is enough to induce a chronic pain state,” said Chen.
The experiments confirmed the circuit’s specific role in chronic pain. “This group of cells is not engaged in normal pain, but only in chronic pain that occurs after injury or inflammation,” Chen said.
The discovery of a new circuit doesn’t mean the previously proposed PAG-RVM pain system is irrelevant. More likely, the two circuits control pain in opposite ways. While stimulating the newly identified circuit increases pain, stimulating the PAG-RVM system reduces pain. “We think that reducing pain and promoting pain are driven by two separate circuits,” said Chen.
Jamming the chronic pain circuit
With the circuit now mapped, Chen is searching for molecular changes that lead the RVM neurons to light up the chronic pain circuit. It may be possible to develop drugs that block this change or that jam the signals the neurons send through the loop. Future medications that target this circuit could potentially ease chronic pain without impacting the ability to feel immediate, acute pain.
Chen is also researching genetic databases from chronic pain patients to determine whether similar changes occur in humans. That work will help researchers confirm whether the same molecular changes identified in mice also control chronic pain in humans, Chen said.
At a deeper level, the discovery raises an intriguing question: Why does a dedicated chronic pain circuit exist at all? Chen thinks it may be linked to how the brain detects internal damage, particularly since the brain itself lacks pain-sensing neurons. For now, “it’s still a mystery,” he said.
Publication details
Research Team
Study authors were Qian Wang, Joo-Han Lee, Gregory Nachtrab, Yuan Yuan, Lei Yuan, Wei Qi, and Manuel Alexander Mohr, and Xiaoke Chen from the Department of Biology in the Stanford School of Humanities and Sciences and Jing Xiong and Mark Horowitz from the Department of Electrical Engineering in the Stanford School of Engineering. Chen and Horowitz are Wu Tsai Neuro affiliates. Chen is a member of Stanford Bio-X. Horowitz is the Fortinet Founders Chair of the Department of Electrical Engineering.
Research Funding
The research was funded by the National Institutes of Health (R01DA045664, R01MH116904, R01HL150566, R01NS129834, R01DA059602, RF1NS126076, and R01MH135934), a Terman Fellowship, a Firmenich Scholarship, the Wu Tsai Neuroscience Institute’s NeuroChoice Initiative, Stanford Medicine and Sarafan ChEM-H’s Innovative Medicines Accelerator, and the Tianqiao and Chrissy Chen Institute. NeuroChoice Initiative funding contributed to generating genetically modified mice used in the study.
Competing Interests
Chen, Wang, and Nachtrab have filed a patent covering some components used in this study (US application: PCT/US2022/07356; Stanford file: S21-124).