Researchers create 'Olympian' mice by stabilizing brain connections involved in skill learning
The idea that a drug could break through the brain's limitations to release our untapped potential has been fodder for many a science fiction tale, but a new study suggests this may not be as far-fetched as you might think. In fact, it could lead to new treatments for neurodegenerative conditions such as Parkinson’s and Alzheimer’s diseases.
When we learn a new skill, whether taking up cross-stitching or practicing a killer serve, the brain must learn to coordinate the dozens or hundreds of muscles involved in the task — what we colloquially refer to as “muscle memory.” In reality, this information is stored in new synaptic connections in the brain’s motor cortex, an arc of brain tissue located about where you might wear a headphone headband. Researchers have shown in animal studies that the memory of a specific skill can even be wiped out by selectively eliminating the relevant connections.
In the new study, published August 25, 2021 in Neuron, researchers with the Wu Tsai Neurosciences Institute at Stanford turned mice into “super-learners” by doing the opposite — stabilizing the new connections formed while animals learned a tricky new movement. This change, which involved inactivating a single protein found at the synaptic connections between brain cells, allowed mice to learn better, faster, and more durably.
The research represents the fruits of a collaboration between the labs of Jun Ding, PhD, which uses cutting-edge techniques to study the brain’s movement planning circuitry, and of Carla Shatz, PhD, which is famed for its seminal work understanding synaptic plasticity — the capacity of brain connections to strengthen and weaken during learning and early brain development.
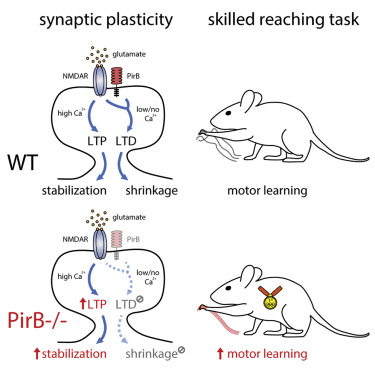
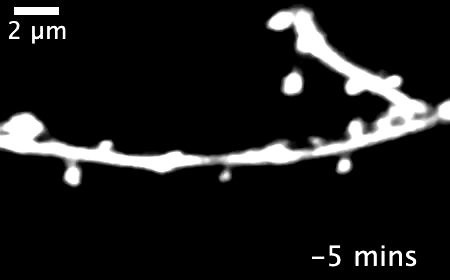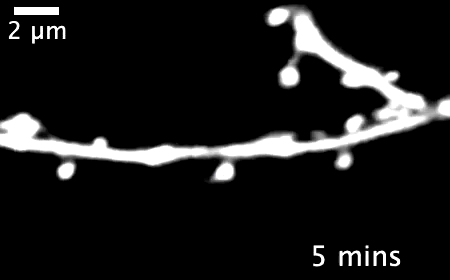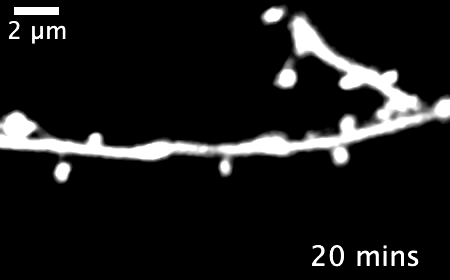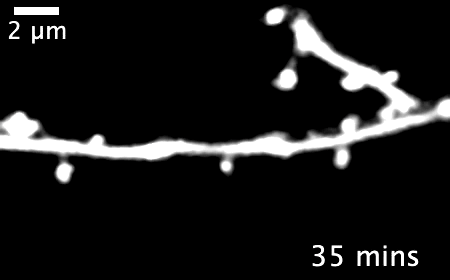
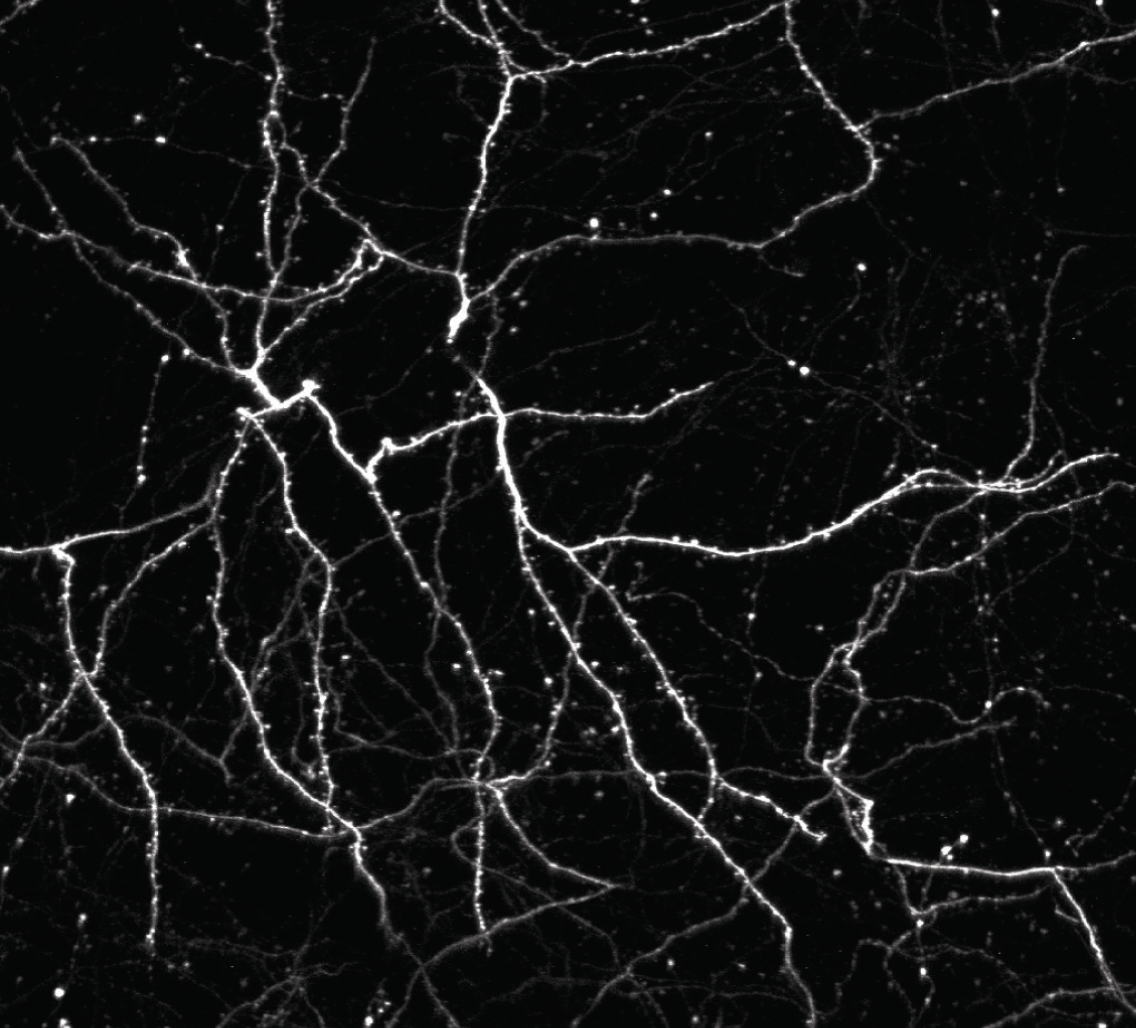
The research revealed that inactivating a synaptic molecule called PIRB in mouse motor cortex stabilized synaptic 'spines' associated with learning and improved the speed and consistency with which animals' learned to reach for a morsel of food. Image credit: Albarran et al, Neuron (2021).
Eddy Albarran, PhD, the study’s lead author, rotated through both labs as a first-year graduate student, and recognized the opportunity to combine the labs’ expertise.
At the time, the Shatz lab had recently discovered that inactivating a single synaptic molecule, called PIRB, can re-open the brain’s remarkable childhood capacity for learning by enhancing the formation of new synapses. And the Ding lab had just discovered that one factor contributing to the loss of fine motor control in people with Parkinson’s disease could be a destabilization of synaptic connections in the brain’s motor cortex.
“Discussing these ideas with Carla and Jun, we started to wonder — if a disease condition impairs skill learning by making synaptic connections less stable, could we also do the opposite — enhancing skill learning by making these connections more stable?” said Albarran, who went on to pursue this question as the nucleus of his dissertation, performed jointly between the two labs.
Deactivating a single synaptic molecule lifts limits on motor learning
The research team began by de-activating PIRB in groups of mice — first genetically and then with a targeted drug — and then presented the animals with a challenge: how to reach through a narrow gap in their enclosures to grasp a millet seed and bring it to their mouths.
Normally, it takes a lot of trial and error for mice to learn how to snag these tasty treats — they often knock the seed away or drop it on the way to their mouths — but with PIRB deactivated, the animals picked up the new skill with remarkable speed.
“I remember one mouse in particular that within five tries had mastered this slow, finessed reach that worked almost every time,” Albarran said. “We were blinded to which animals were which during the experiment, but this animal was clearly gifted. It was getting the seeds at least twice as often as the average mouse.”
These “super-learners” not only performed better from day one, but they maintained this advantage over their siblings with intact PIRB when researchers tested them again a month later.

Mice learn to grasp a tasty millet seed through trial and error. Animals with a single synaptic molecule disabled mastered the task much faster, often within five tries. Image credit: Eddy Albarran, PhD.
“What I think is amazing about this experiment is that we could take normal adult mice, and with just a brief, 10-day intervention with a targeted drug, turn them into Olympians. We changed the animals’ physical performance just by altering their brains,” said Shatz, who is Sapp Family Provostial Professor in the departments of Biology and of Neurobiology, Catherine Holman Johnson Director of Stanford Bio-X, and a member of the Wu Tsai Neurosciences Institute.
“There are all kinds of ways to impair an animal's learning and performance, but it’s very rare to see a pharmaceutical manipulation that can lift a limit and enhance learning,” added Ding, who is an associate professor of neurosurgery and of neurobiology, and a member of Stanford BioX and the Wu Tsai Neurosciences Institute.
Intervention restores child-like learning abilities by blocking synapse weakening
The speed and stability of skill learning in these “Olympian” mice was also reflected at the level of the animals’ brain circuits, which Albarran and colleagues studied by peering through surgically implanted windows in the animals’ skulls with high resolution microscopes capable of visualizing individual synaptic connections.
Normally, learning a new skill causes many new synapses to form in the motor cortex, followed by a period of synapse loss — keeping the total number of neural connections relatively stable. But in animals without functioning PIRB, the new connections formed during learning rarely went away and could still be found more than 30 days after the initial training.
PIRB appears to be required to weaken less-used synapses, the researchers concluded, part of a constant push-and pull mechanism called activity dependent plasticity, which normally keeps brain circuits well-pruned by retaining only the strongest, most relevant synaptic connections.
Albarran showed how the loss of PIRB tips this balance in a delicate experiment in which he stimulated individual synapses with precisely timed release of the neurotransmitter glutamate. Normally, this would allow him to trigger activity-dependent plasticity rules to either strengthen or weaken individual synaptic connections. But in the brains of animals without functional PIRB, synaptic connections once strengthened by learning could no longer be weakened by disuse, resulting in more rapid and stable learning.
The researchers did note some potential downsides to this enhanced synaptic plasticity in mice lacking PIRB. Specifically, super-learners were quicker to produce consistent, stereotyped reaches than unmodified animals that learned the task more slowly. Though more research is needed on the question, this suggested to the researchers that an important role of PIRB may be to shift the brain from a childhood state of rapid, long-lasting learning — like learning to speak or how to ride a bike — to an adult’s slower but more flexible learning abilities.
But the researchers are eager to pursue the new findings as a potential approach to restoring function in people with disorders that drive synaptic destabilization.
“This has been a wonderful convergence of our labs — driven by neurodegeneration on one hand and neuroplasticity on the other,” Shatz said. “Our hope is to take the lessons we’ve learned from the young brain about why kids learn so rapidly and adults don't, and combine it with what we are learning about the loss of plasticity in neurodegenerative disorders like Parkinson’s and Alzheimers, to eventually lead to interventions that can potentially restore function and improve quality of life.”
Additional Stanford authors on the study were postdoctoral fellows Aram Raissi, PhD, and Omar Jáidar, PhD.