Q&A: Reverse engineering the human brain by growing neural circuits in the lab
Neuroscientists face a paradox.
The field aims to understand the mysteries of the human mind, but studying the actual human brain cells and circuits that produce our mental lives—and how they go awry in neuropsychiatric disease—is incredibly challenging. After all, the brain is packed away carefully in our skulls, and people typically don’t part with samples of their brain tissue lightly.
Over the past decade, Wu Tsai Neurosciences Institute faculty affiliate Sergiu Pasca and his team of collaborators have pioneered transformational new techniques to grow simplified human brain circuits in the lab from human stem cells, allowing them for the first time to study the assembly and wiring of the human brain — and the molecular rules that orchestrate it — in real time.

Sergiu Pasca, Uytengsu Family Director of the Stanford Brain Organogenesis Program at the Wu Tsai Neurosciences Institute. Image credit: Steve Fisch.
Taking advantage of recent advances in stem cell technology, the team can now “reboot” skin cells from a healthy research subject or a patient with a neuropsychiatric disorder, then guide the resulting stem cells to develop into various types of brain cells. Using molecular cues discovered by Pasca’s lab, the team can now produce over a dozen different types of brain and nervous system tissues, grown in laboratory dishes as floating spheres of cells called “organoids.” Organoids recapitulating different brain regions can then be combined to grow simplified human brain circuits, dubbed “assembloids.”
Researchers around the world are now using brain organoid and assembloid techniques to turn back the clock on human brain development: to directly observe the rules governing the self-assembly of our unique neural circuitry and to pinpoint how the process goes awry in brain disorders such as ALS, autism or schizophrenia—in hopes of identifying targeted therapies for these conditions.
Lab-grown neuronal organoids allow scientists to study how different parts of the developing brain wire together. Image courtesy Pasca Lab.
To advance and share these techniques, Pasca, an associate professor of psychiatry and behavioral sciences, and Stanford optogenetics pioneer Karl Deisseroth have convened psychiatrists, neuroscientists, engineers, chemists, and bioethicists from across Stanford to form the Stanford Brain Organogenesis Project, funded by a Big Ideas in Neuroscience award from the Wu Tsai Neurosciences Institute. As the Uytengsu Family Director, Pasca also leads a collaborative laboratory space in the new Stanford Neuroscience Building dedicated to serve as a hub for the growing worldwide field he helped launch.
Organoids and assembloids were recently hailed as "the leading edge of stem-cell research" in the journal Nature, and in November Pasca and lab member Kevin Kelley published a detailed overview of the techniques for those looking to get into the new field.
We spoke with Pasca about the origin of this new field, and its potential to reveal new insights into the rules of biological self-organization that produce our ability to think, feel, plan and dream.
There’s a critique out there that neuroscientists have “cured” Alzheimer’s, ALS and other brain diseases dozens or hundreds of times in rodents, but these advances have so far not led to human therapies. Why is it critical to develop new platforms specifically for studying human brain tissue and brain development?

An animated summary of Pasca and Kelley's Nov, 2021 Cell review of the neural organoid and assembloid field. Courtesy Kevin Kelley, Pasca Lab.
Well, most of what we know about how the human brain develops is by extrapolation from studies in rodents. There's no doubt that animal models have been incredibly useful, but at the same time, it’s no surprise that the human brain is quite unique from the rodent brain. There are about 70 million years of evolution that separate us. There are certain cell types that exist only in the human brain and our brain connectivity is often also different. If we want to understand neuropsychiatric disease, the specific human genomic context also matters a lot: for example, the same genetic deletion can produce very different conditions from one person to another because of human-specific genomic context that is hard to study in a rodent.
We need to be able to work with human brain tissue and specifically with patient-derived tissue if we want to understand these uniquely human neuropsychiatric disorders. Fortunately, a number of technological advances across several fields are now making it possible to model aspects of brain development relevant to a specific person with psychiatric disease in a non-invasive way and to profile with high accuracy cell diversity and cell states in actual human brain tissue. It feels as though we are at an inflection point, which motivated writing this overview of the field for Cell.
Do you think people outside the field would be surprised about how little we know about the specifics of human brain development and the origins of developmental brain disorders?
Absolutely. It turns out the further along you look in human brain development, the less we know. We know quite a lot about early stages—how the neural tube develops and starts to specialize—but we previously have not been able to comprehensively study human brain tissue during the second and third trimester and soon after birth. Those stages, when human disease can often arise, remain, to a large extent, uncharted.
This is important because a very salient feature of human brain development is that it's incredibly long. Just to give a couple examples, newborn neurons continue to migrate across the brain in humans into the second year of life, and myelination of nerve fibers continue into the second and third decade! We need new ways to study what is going on during these later periods of brain development — for instance how some of the unique cell types seen only in humans are produced, how the wiring of the human nervous system has been adapted evolutionarily and why our brain takes so long to mature. There are likely way more unknowns about human brain development that we haven’t even guessed at.
Can you tell us about how you came to the idea of brain organoids to address this problem?
Neural organoids in a laboratory dish. Image courtesy Pasca Lab.
We published the first study reporting the derivation of neurons from stem cells in a genetic form of autism 10 years ago. At first, we were only able to keep these human neurons alive in a dish for 10–12 weeks, and that was through heroic efforts. That’s nothing in the context of human brain development: It takes about 27 weeks just to make all the neurons in the human cerebral cortex!
So that’s why, around 2011, we started playing with the simple idea of performing the neural differentiation not at a bottom of a dish, but in low-attachment plates where cells could grow in suspension. And it turned out that this new degree of freedom unleashed surprising self-organization forces and we could ultimately keep these three-dimensional clusters of neural cells indefinitely. We've maintained some of the longest cultures that have ever been reported—going beyond 800 days or so. So that was the original motivation for developing what are now known as self-organizing neural organoids—just trying to keep patient derived cells alive longer in the lab.
Since discovering this ability to differentiate stem cells in 3D, we have been working to identify instructive signals to guide them to recapitulate the development of different human brain regions. We can now produce organoids of more than a dozen distinct brain regions, such as cortex, spinal cord, striatum, or thalamus. And then of course the next question was, “Can we put them together? And will they join up in a way that is reminiscent of what takes place in utero?” “Can we build functional human circuits in a dish?”
Once you've got these organoids that recapitulate a particular piece of the brain, what are some of the questions that you can start tackling by combining them into ever more complex assembloids?
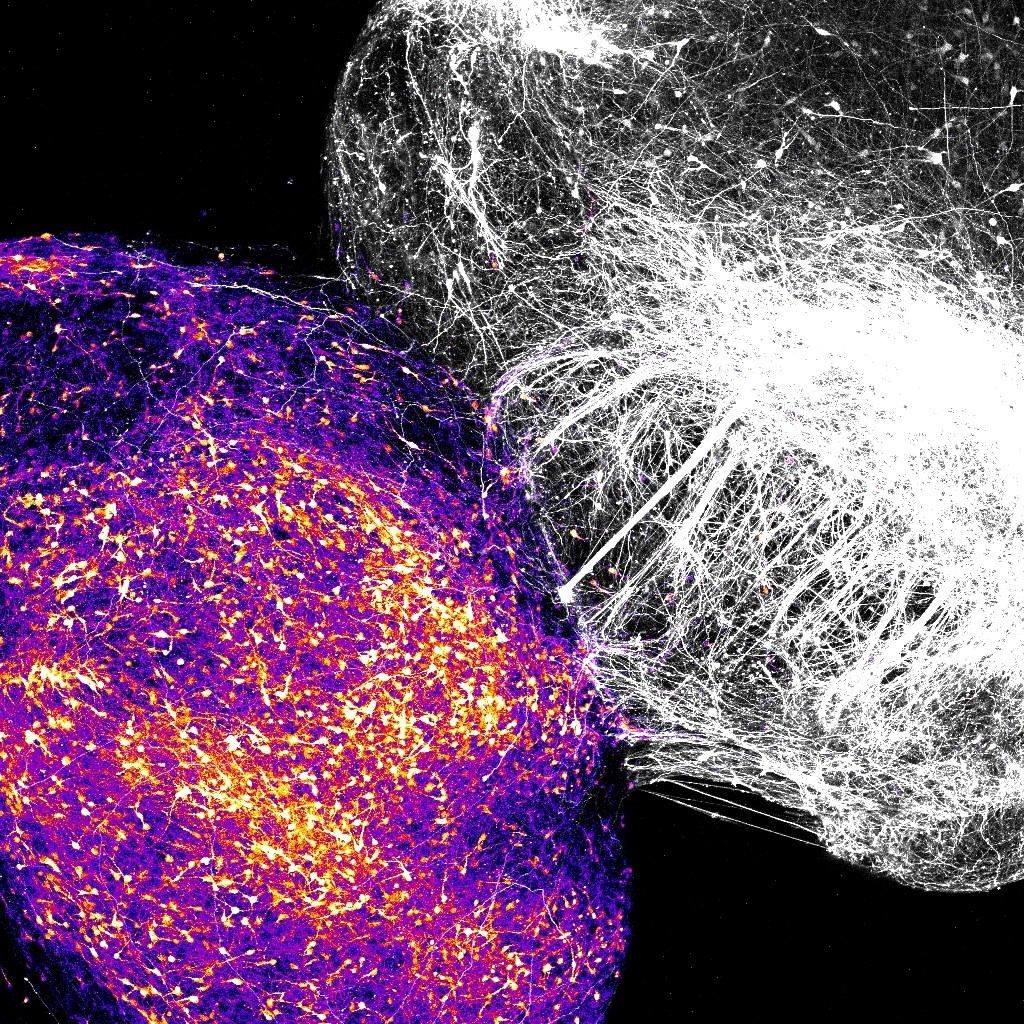
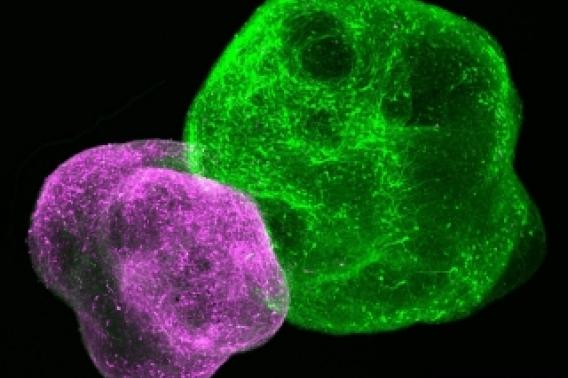
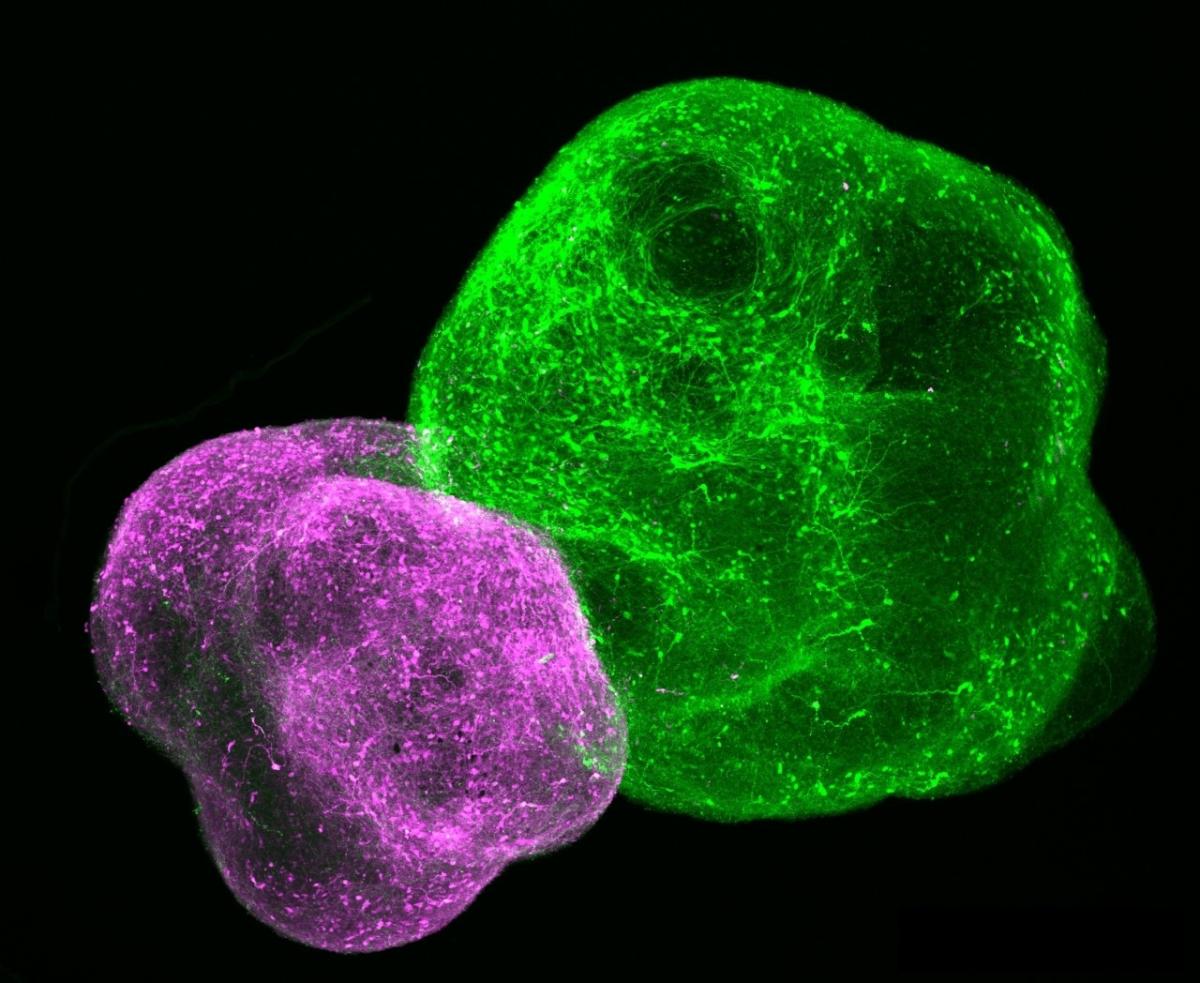
Neural organoids representing striatum and cortex merge in a laboratory dish, enabling scientists to study the rules governing the growth of human brain circuits. Image courtesy Pasca Lab.
This was a very early dream of mine, even before I started my lab in 2014. We weren’t yet at the stage of making many different brain regions, but I knew that to model the development of human circuits, we would need to find a way of putting these different organoids together. At first I worried that it was going to be very difficult to get these organoids to fuse with each other. We spent months thinking about how we’d use nano-electrodes or other devices to convince them to link up. But in the end, it turned out to be as simple as leaving them together in an Eppendorf tube overnight. Next morning, they were fused.
Not only did the cells recognize each other, but they started to self-organize in ways that resemble human brain development. In our very first assembloid, we fused ventral forebrain and dorsal forebrain (cortex) organoids and right away saw newborn GABA-ergic interneurons start to migrate towards the cerebral cortex, which is a unique and important aspect of human brain development.
Since then, we’ve put together cortex and striatum organoids, and see that cortical cells quickly send out projections to striatum but not the other way around, which is just what you see in an intact brain. In the most advanced assembloid we published so far, we put together cerebral cortex, spinal cord and muscle organoids, and get beautiful projections from specific types of cortical neurons to spinal cord neurons, which then project to muscle cells such that we can even trigger muscle contraction using optogenetics in a multi-synaptic circuit.
What has this taught you about the rules guiding brain development?
I think there's a very important lesson out arising from these experiments. When I was being trained as a physician, we were taught to think like mechanical engineers, with the human body as a machine. So my assumption was always that if we wanted to build a circuit of the brain, we’d better know all the rules for that assembly process: why this cell projects to that cell, and be able to provide the necessary guidance.
But that perspective forgets that what we're dealing with is not an engineering problem, but a biological self-organizing system. The human brain builds itself. And so when you make these brain regions separately and then combine them, some of the rules for assembly come with the system. Neurons find each other and activate subsequent programs of maturation and assembly. As a result, you’re not just recreating the process that you planned to model, but you have a chance of getting a glimpse at the deeper interactions in the system that you may not have expected.
Learn More: Miura et al. "Engineering brain assembloids to interrogate human neural circuits." Nature Protocols, January 6, 2022.
I think that in itself is an absolutely superb opportunity to now start to reverse engineer some of these rules in the lab: Why do some cortical neurons only project to the striatum while others only project to the spinal cord? What aspects of interneuron migration are unique to the human brain, and how might they be affected in neuropsychiatric disease?
What are some of the questions about the development of neuropsychiatric conditions such as schizophrenia or autism that you are most excited to model, and that have a chance of leading to new understanding of these conditions?
For both schizophrenia and for autism, to use your examples, there is evidence that cellular and molecular processes taking place in mid- and late gestation are important factors in disease risk. Most of the genes that are associated with these disorders are expressed in that period and we know from epidemiological studies that, for instance, maternal viral infection in the third and second trimester come with a higher risk for developing disease.
Learn More: Birey et al. "Dissecting the molecular basis of human interneuron migration in forebrain assembloids from Timothy syndrome." Cell Stem Cell, Jan 05, 2022.
This makes sense because in humans, this period of late gestation is a crucial time for generating very specialized cell types, for migration of neurons from where they’re born into their proper circuits, and for establishing the connectivity between brain circuits. Anything that disrupts these processes during this sensitive time could contribute to development of these disorders.
With organoid and assembloid models derived from patient stem cells, we can study these processes at an equivalent developmental stage to this critical period of late gestation — exactly the time period that had been so difficult to study at a mechanistic level before. I think it’s a very exciting time — as I’ve said, it’s still early days, but we now have the platform to study these questions in a very detailed way in a human-relevant context and this brings an opportunity for transformational new insights.
Assembloids allow researchers to study the formation of human brain circuits in action.
Video courtesy of Yuki Miura, Pasca Lab.
Your Stanford Brain Organogenesis Program is supported by a Wu Tsai Neuro Big Ideas in Neuroscience award, which emphasizes promoting interdisciplinary collaborations to help advance the field. How have you been able to bring together people coming from different areas of science and engineering to advance these models?
Early on, we recognized that even once we could reliably create specific organoids and assembloids, we didn't have the necessary tools to study them. Optogenetic stimulation or large-scale electrophysiological recordings had not been applied to 3D clusters of human neurons at that point. Delivering specific viruses to human cells to probe and manipulate cells was also challenging. Fortunately, we are so at the forefront of neurotechnology development here at Stanford, I was able to go around and discuss these needs with the pioneers of these technologies and develop our Stanford Brain Organogensis Program with support from the Wu Tsai Neurosciences Institute.
My first and very close collaborator was Karl Deisseroth, who pioneered optogenetics, CLARITY, and many other methods that were essential for studying these organoid models. Then we expanded to include people outside of the medical school, including Zhenan Bao, Bianxiao Cui, Sarah Heilshorn and Michael Lin, each of whom have developed unique technologies to advance these models from different angles, such as extracellular matrices to support developing organoids or flexible electronics and voltage sensors for recording from the cells. We also recognized that there are a number of ethical and legal issues that arise from this type of studies, so we were very lucky to have bioethicist Hank Greely from Stanford Law involved in these conversations from very early on.
It has been a deeply rewarding experience for all of us. One of my favorite aspects has been the series of cross-disciplinary meetings we held with all our collaborators and their students in the Gazebo near the chemistry building. It was amazing to see the incredible brainstorming energy we produced at these “Gazebo meetings” just by getting chemistry students, engineering students, and hardcore neurosciences students all in the same room, which has launched ideas and collaborations that never would have happened otherwise.

Members of the Stanford Brain Organogenesis Program, a Big Ideas Initiative supported by the Wu Tsai Neurosciences Institute. From left to right, Karl Deisseroth (D. H. Chen Professor of Bioengineering and of Psychiatry and Behavioral Sciences), Michael Lin (Associate Professor of Neurobiology & of Bioengineering), Zhenan Bao (K. K. Lee Professor of Chemical Engineering), Sarah Heilshorn (Professor of Materials Science and Engineering), Bianxiao Cui (Job and Gertrud Tamaki Professor of Chemistry), Hank Greely (Deane F. and Kate Edelman Johnson Professor of Law), and Sergiu Pasca (Bonnie Uytengsu and Family Director of the Stanford Brain Organogenesis Program and Associate Professor of Psychiatry and Behavioral Sciences). Photo credit: Avery Krieger.
How have you been utilizing the collaborative Brain Organogenesis Program laboratory in the Stanford Neurosciences Building to advance this technology?

Students in the first Stanford Brain Organogenesis course at the Wu Tsai Neurosciences Institute. Image courtesy Pasca Lab.
One of the things that makes Stanford unique is that we're one of the only top medical schools that is within a walking distance from an engineering school, not to mention departments of chemistry, biology, genetics at so on in the School of Humanities and Sciences. The new space we have in the Wu Tsai Neuroscience building, strategically located at this nexus of collaboration, has been an incubator of ideas where students from different labs and disciplines can come together to do experiments and learn from one another. Already, many Stanford labs have been applying this technology in various ways, such as in Tony Wyss-Coray and Aaron Gitler’s Big Ideas in Neurosciences initiative to study brain rejuvenation and aging, William Greenleaf’s work studying chromatin dynamics in brain development or Jan Carette’s work developing human cellular models of enterovirus infection.
It has been important for us from the beginning to make this technology broadly accessible—in part because there are just so many more potential applications than we could do ourselves. So we launched a new hands-on course, which we designed kind of like a cooking show, where we go through the key steps of creating human organoids and assembloids and thinking about how to use them to model disease. There was tremendous interest in our first course from students around the world — we had over 200 candidates for 20 spots. We ultimately hosted students from four continents for a full week of this immersive experience, all of whom pledged to will go back home and teach others. As a result, there are now people using this technology all over the world — not only here in the US but also in Portugal, Chile, South Korea, Australia, and so on. I think this has been incredibly successful in amplifying Stanford as a hub for this new technology and the study of human neurobiology.
Where do you hope this technology is in another 10 years?
There are so many aspects of human brain development that are still inaccessible, such as advancing maturation of cells to study neuro-degeneration and enabling studies of plasticity of brain circuits. Through some of our collaborations, we’ve been building some next-generation models to address these questions. This is going to be a long journey, but I think this is the beginning of a new era of molecular psychiatry, moving from a largely behavioral understanding of the human brain to a mechanistic understanding of mental health and disease.